
WOLBACHIA BACTERIA IN ACTION | How we’re using naturally occurring bacteria to stop mosquitoes from spreading disease
Words and images: Perran Ross
Cover photo: Jason Axford
Dengue is a major global health issue. It infects millions of people every year and can cause debilitating illness, inflicting joint pain, rash and fever. Without any effective vaccine, the best way to prevent dengue is to target the mosquitoes that transmit it. Dengue is spread by Aedes mosquitoes, and we’ve been waging war on them for decades with an arsenal of repellents, traps and insecticides. But mosquitoes have put up a fight. Aedes aegypti, the main vector of dengue, is becoming resistant to many insecticides and this will make it very difficult to control mosquito populations in the long term.
But is there a way to prevent dengue transmission without killing mosquitoes? Enter Wolbachia.

Aedes aegypti (left) is the main vector of dengue virus (right)
Wolbachia are bacteria that occur naturally inside the cells of insects. They are perhaps the most prolific symbionts on earth, infecting up to half of insect species. The secret to their success can in part be explained by their ability to hijack the reproduction of their hosts. Different types of Wolbachia have different tricks up their sleeve, usually resulting in an increased production of females (which transmit Wolbachia to their offspring) at the cost of males (which do not transmit the infection). Wolbachia can turn males into females through feminisation, remove the need for males entirely through parthenogenesis, or just kill males outright! Some of these bacteria can be used to reduce the size of mosquito populations.
But not all Wolbachia are so selfish. Some strains can be beneficial, giving their hosts direct advantages by increasing the number of eggs they lay, or by protecting them from pathogens. It’s this feature that makes it a particularly promising tool for mosquito-borne disease control. By introducing Wolbachia into Aedes aegypti mosquitoes, we could reduce their ability to transmit dengue and other nasty illnesses.
Several types of Wolbachia have now been introduced into Aedes aegypti by injecting the bacteria from other insects into mosquito eggs. Once a female mosquito is infected with Wolbachia, she will pass the infection to all her offspring. But how do we get these mosquitoes into wild populations?
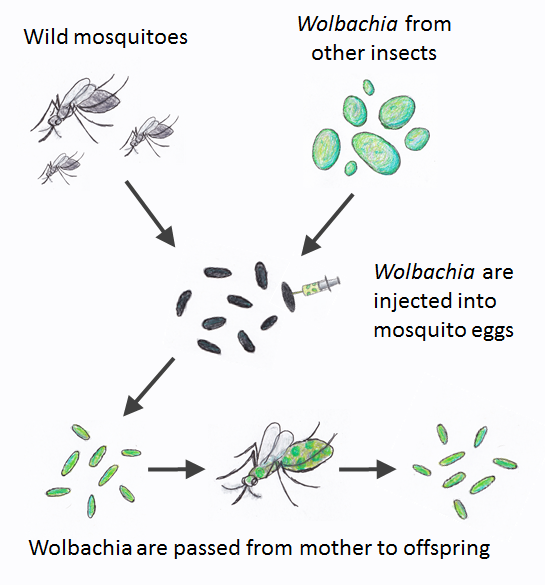 Wolbachia can be taken from other insects and introduced into disease transmitting mosquitoes
Wolbachia can be taken from other insects and introduced into disease transmitting mosquitoes
When introduced into mosquitoes, Wolbachia causes cytoplasmic incompatibility. This means that when males infected with Wolbachia mate with females that are not infected, their eggs do not hatch. This gives females infected with Wolbachia the upper hand, assisting the spread of Wolbachia into wild mosquito populations.
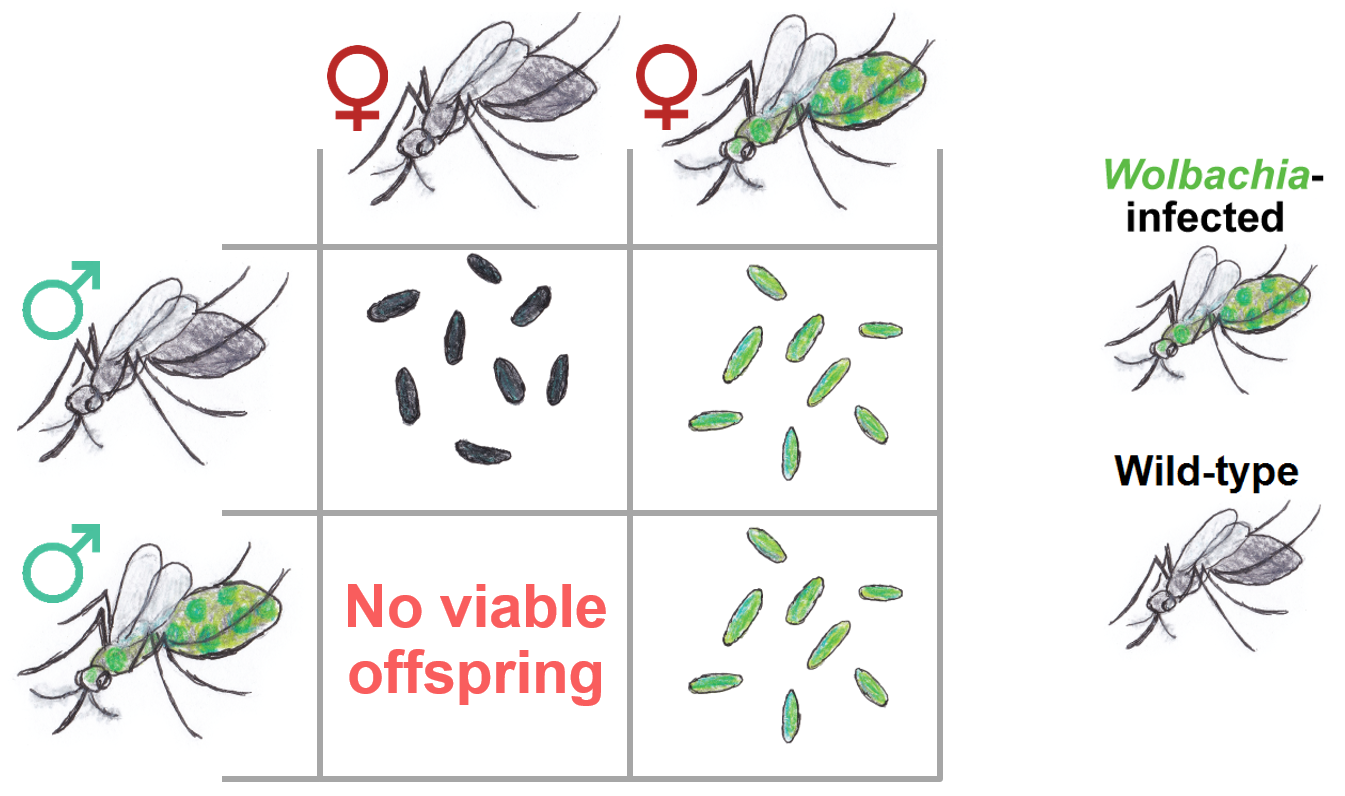 Wolbachia give female mosquitoes a reproductive advantage
Wolbachia give female mosquitoes a reproductive advantage
But how does Wolbachia to stop disease transmission? As it turns out, not only do Wolbachia protect their native hosts from pathogens, but they also protect mosquitoes from viral infections. Wolbachia reduce the ability of Aedes aegypti mosquitoes to transmit dengue, Zika, Chikungunya and other viruses, making them ideal tool for controlling disease transmission without needing to eradicate mosquitoes. In addition, the Wolbachia that reduce the size of mosquito populations also further decrease the transmission of dengue and other viruses.

Wolbachia block dengue transmission
Mosquitoes with different Wolbachia strains have now been released in Australia and several countries in Asia and South America where they have established in the wild. In the first trials in northern Queensland, Wolbachia invaded populations very rapidly and has stayed there for over seven years.
These field releases are promising, but there is still a lot of work to be done. We need to carefully monitor areas where Wolbachia have been released and assess its impact on disease transmission, and characterise new Wolbachia strains that could be even more effective at stopping dengue. It may be possible to release Wolbachia strains that not only reduce transmission but also decrease the size of mosquito populations. Wolbachia cause a diversity of effects on their insect hosts, and there are many ways in which these bacteria and other types of bacteria might be used in reducing disease transmission.
 Wolbachia are found in up to half of the world’s insect species including mosquitoes, aphids and tiny parasitoid wasps
Wolbachia are found in up to half of the world’s insect species including mosquitoes, aphids and tiny parasitoid wasps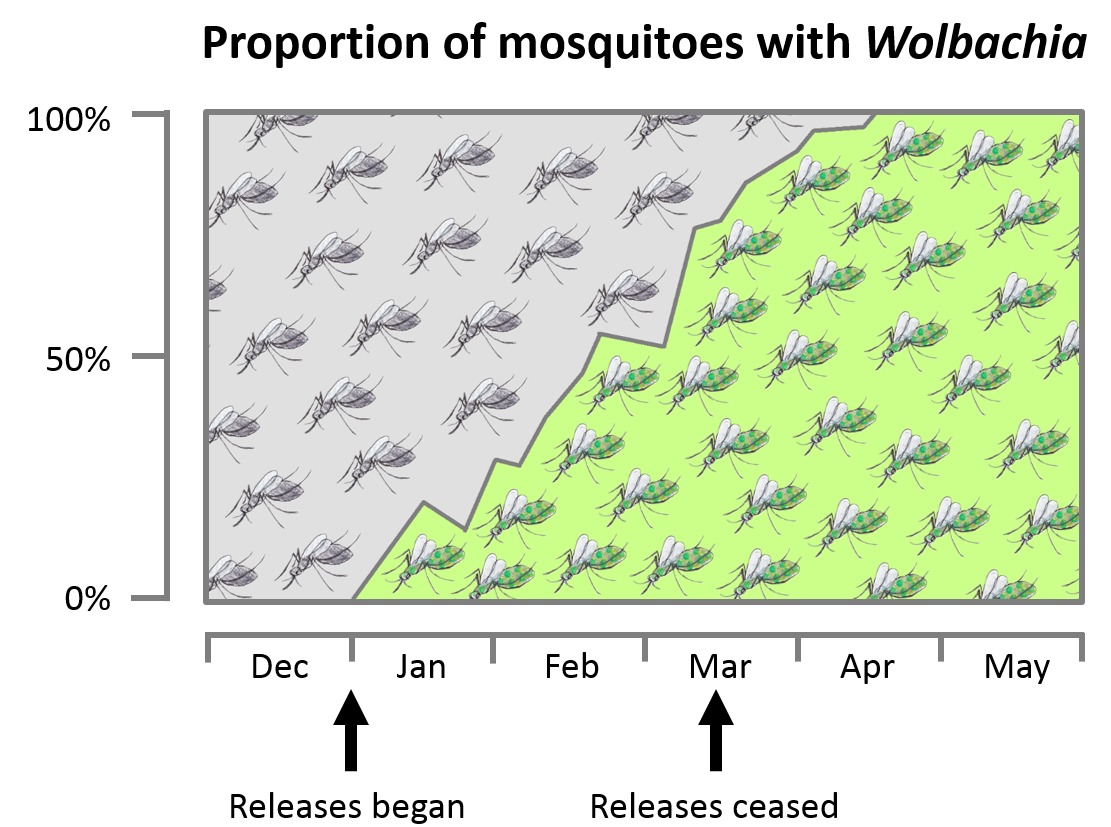 Some Wolbachia strains can rapidly invade natural mosquito populations, although others can be more difficult to spread
Some Wolbachia strains can rapidly invade natural mosquito populations, although others can be more difficult to spread